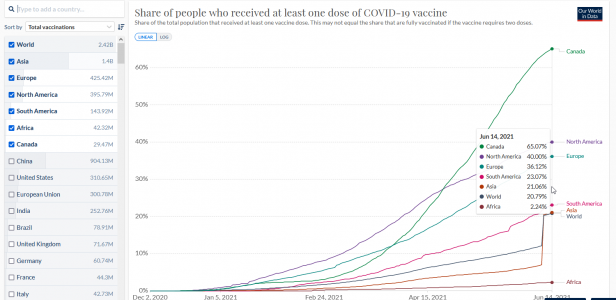
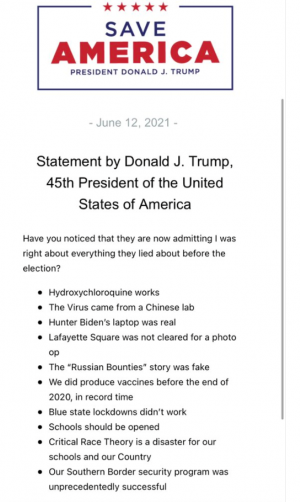
美国打得已经死了5000了,这数据我害怕
cdc Vaers 12月14日到5月28日 5165人,重伤 25359人,不良反应 294801.
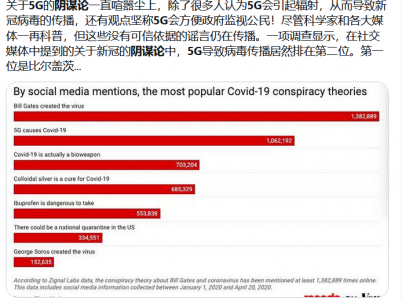
哈哈,也许假的也不一定啊,毕竟阴谋论满天飞。
我自己只是比较谨慎而已

Vaccine Adverse Event Reporting System (VAERS)
VAERS is a warning system used to monitor events after vaccination.
How reports come into VAERS
VAERS collects reports of possible adverse events that happen after vaccination. As a condition of a vaccine’s use under Emergency Use Authorization, the FDA requires healthcare professionals to report to VAERS certain adverse eventsexternal icon that occur after COVID-19 vaccination.
However, anyone can submit a report to VAERSexternal icon, including patients, family members, healthcare providers, and vaccine manufacturers, even if it isn’t clear if the vaccine caused the health problem.